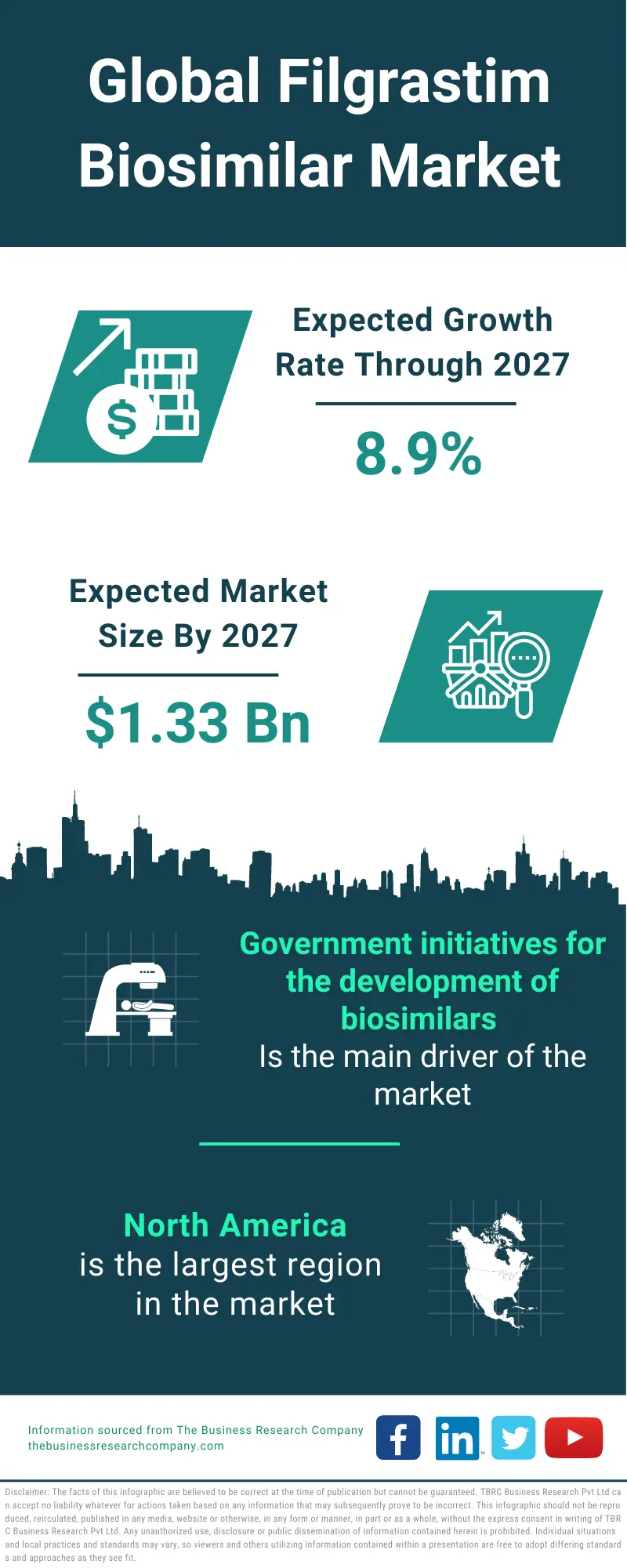
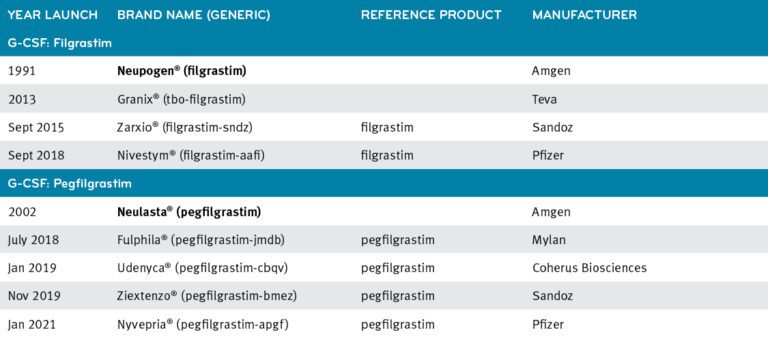
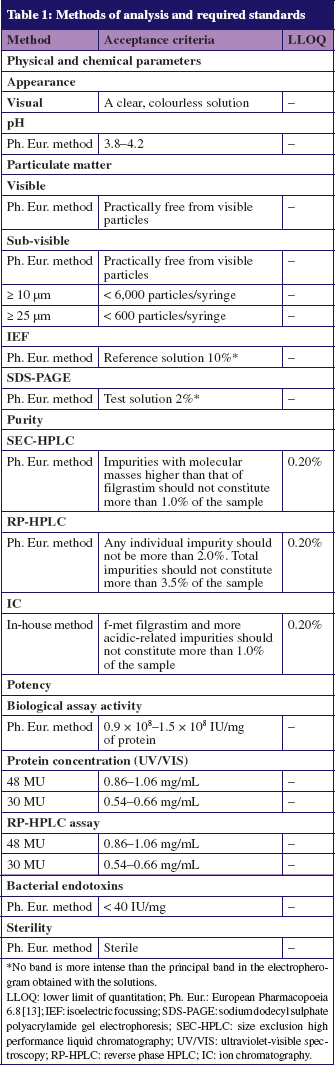
Key concepts and critical issues on epoetin and filgrastim biosimilars. A position paper from the Italian Society of Hematology, Italian Society of Experimental Hematology, and Italian Group for Bone Marrow Transplantation

These highlights do not include all the information needed to use GRANIX safely and effectively. See full prescribing information for GRANIX.GRANIX® (tbo-filgrastim) injection, for subcutaneous use Initial U.S. Approval: 2012

Tevagrastim - Filgrastim 300mcg/0,5ml - Solução Injetável - Teva - ÁgilMed - Medicamentos Especiais e Nutrição Clínica